Crowley1 project protocol
Drug study: Haloperidol-induced vacuous chewing movements, extrapyramidal side effects, and spontaneous locomotor activity in males of 27 inbred mouse strains (2012)
Crowley JJ, McLeod HL, Sullivan PFWith: Adkins DE, Pratt AL, Quackenbush CR, van den Oord EJ, Moy SS, Wilhelmsen KC, Cooper TB
Project protocol — Contents
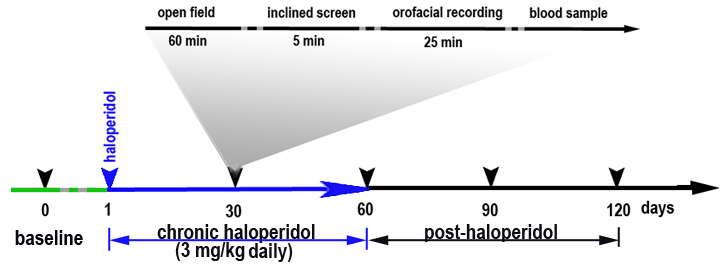
Workflow and sampling
Equipment
Reagents, supplies, and solutions
Procedures 1: Haloperidol administration
Procedures 2: Battery of behavioral tests
Procedures 3: Plasma haloperidol concentration
Data
Definitions
ReferencesWorkflow
(haloperidol)Baseline (pre-treatment) testing: locomotor activity evaluated in the open field; extrapyramidal side effects assessed using inclined screen test; orofacial behavior recorded on mice in restrictor device under anesthesia with a high-resolution digital video camcorder Analysis: involuntary orofacial behavior
- Balance scale
- Precision trocar for drug pellet delivery
- Photocell-equipped automated open-field apparatus (Versamax system, Accuscan Instruments, Columbus, OH, USA; 40 cm wide x 40 cm long x 30 cm high; center 20 x 20 cm; (Fig. 2), placed inside sound-attenuating boxes equipped with houselights and fans
- Wire mesh screen (~0.5 cm2) inclined 45°, approximately 34 cm long x 11 cm wide (Fig. 3)
- Custom made horizontal platform with collars composed of two semicircular pieces of plastic: a lower piece fixed to the platform and an upper piece used as restrainer around the neck; both the diameter of the collar and its height above the platform are adjustable allowing for a comfortable posture during video recording (Fig. 4)
- High-resolution digital camcorder (JVC Everio GZ-MG360BU; Wayne, NJ, USA) for recording orofacial behavior (Fig. 4)
- Observer video analysis system (Noldus, Wageningen, The Netherlands)
- Mass spectrometry by the Analytical Psychopharmacology Laboratory at the Nathan Kline Institute for Psychiatric Research (Orangeburg, NY, USA)
Reagents, supplies, solutions Â
- Haloperidol pellets (Innovative Research of America; Sarasota, FL, USA)
- Isoflurane gas anesthesia setup
- Disposable blades
- Blood sample collection tubes with anti-coagulant
- Paper towels
- Cleaning solution and disinfectant
Procedures 1: Haloperidol administration
a. Pilot studies indicate that a haloperidol dosage of 3.0 mg/kg body weight per day delivers a steady-state plasma concentration in the target range (10-50 nM, human-like steady state concentration). Therefore, this regimine delivers a steady, chronic haloperidol exposure through day 60 post-implantation; lasting drug effects can be studied beyond day 60.
b. Each mouse is weighed and a 60-day release implant is formulated from engineered pellets ranging from 2 mg to 10 mg (in 0.5 mg incremements) to deliver the required dosage. Before receiving an implant, mice are anesthetized with isoflurane for at least 2 min to minimize handling stress and pain.
c. Haloperidol pellets are implanted subcutaneously along the cervical region using a trocar (Note: For the first few days after exposure to haloperidol, food and water are positioned more accessibly near the bedding surface).Procedures 2: Battery of behavioral tests
Behavioral measurements are obtained with the least invasive test first and culminating with the collection of tail blood samples (see below). Testing is done at specified times post-implantation (Fig. 1).
1. Open field test
a. Mice are placed in the open field arena (Fig. 2) and monitored for spontaneous locomotor activity via built-in photocells for a duration of 1 h.
b. Recorded activity data are parsed according to total distance traveled (cm), vertical activity (number of beam breaks), stereotypy (repeated breaking of the same beam, number of beam breaks), and time spent in the central region of the arena.
c. Mice are tested on days 0 (baseline), 30, 60, 90, 120 (relative to drug implant on day 1).
e. The open field arena is thoroughly cleaned after each test.
2. Inclined screen test
a. Mice are placed in the middle of the screen with their heads toward the top and their tails down (Fig. 3).
c. The latency to move all four paws is recorded for maximum duration of 5 min.
d. Mice are tested on days 0 (baseline), 1, 2, 3, 4, 5, 6, 7, 30, 60, 90, 120 (relative to drug implant on day 1).
e. The inclined screen setup is thoroughly cleaned after each test.
3. Orofacial behavior assessment
a. A custom made restrictor device is assembled atop a horizontal platform (modified from the method of Tomiyama et al.) equipped with high-resolution digital camcorder properly positioned for recording orofacial movements (Fig. 4).
b. Each mouse is briefly anesthetized using isoflurane and placed in the restrictor device.
c. With the mouse partially immobilized around the neck, a high-resolution digital camcorder is focused onto the orofacial region from below the horizontal platform.
d. Each mouse is kept in the restrictor device and video recorded for 25 min.
e. The video recording platform setup is thoroughly cleaned after each use.
f. Fully trained raters*, blinded to all study data including strain and study time point, are then randomly assigned to score the final 15 min of each video recording.
g. The final 15 min are scored according to four distinct orofacial movements: tongue protrusions, overt chewing movements, subtle chewing movements, and jaw tremors. Tongue protrusions and chewing movements are measured as counts of individual events and tremors as total duration.
h. Mice are tested on days 0 (baseline), 30, 60, 90, 120 (relative to drug implant on day 1).* Behavioral raters are trained by an experienced rater in a three-step process:
1. Viewing of a training tape with clips from several different strains of mice demonstrating each movement phenotype (tongue protrusion, overt and subtle chewing movements, jaw tremors);
2. Comparative scoring of a training set composed of 36 video clips (one haloperidol and one placebo treated mouse from 18 different strains);
3. Scoring of training tapes continues until the new rater reaches consensus criterion. To monitor drift, the same training tape is periodically and blindly scored.Submitting PI notes: "Inter-rater reliability among the three raters was high (intra-class correlation of 0.92), as determined by individual scoring of 32 randomly assigned and blinded video recordings."
Procedures 3: Plasma haloperidol concentration
a. After conducting behavioral tests, a tail blood sample is collected from each mouse in the presence of anticoagulant.
b. Blood samples are centrifuged for at least 5 min to separate the plasma.
c. Plasma samples are kept frozen until assayed using mass spectrometry.
d. Plasma haloperidol concentration is determined on samples taken on days 30, 60, 90, 120 (relative to drug implant on day 1).
Figure 2. Open field apparatus.
Figure 3. Inclined screen test.
Figure 4. Restrictor device for recording orofacial behavior.Data collected by investigator
- body weight
- center occupancy in an open field
- repetitive behavior (stereotypy) in an open field
- distance traveled in an open field
- vertical activity in an open field
- tongue protrusion
- latency to move on an inclined screen test
- overt chewing movement
- subtle chewing movement
- jaw tremor
- plasma haloperidol concentration
acute extrapyramidal syndromes (EPS): restlessness marked by inability to remain still (akathisia), sustained muscle contractions (dystonia), and secondary parkinsonism (tremor)
late-onset (chronic) extrapyramidal symptoms (EPS): may develop in subjects treated chronically with antipsychotic drugs; characterized by repetitive, involuntary and purposeless movements primarily of the orofacial region (for example vacuous chewing and tongue protrusion); may or may not be reversed after drug exposure)
tardive dyskinesia (TD): clinically relevant, late-onset EPS
vacuous chewing movements (VCMs): phenotypically and pharmacologically valid animal model of TD
haloperidol: typical antipsychotic drug used in the treatment of schizophrenia, as well as in the treatment of acute psychotic states and delirium
total distance traveled: total horizontal activity or locomotion
vertical activity: number of vertical beam breaks
stereotypy: repeated breaking of the same beam
chewing movement: defined as a single mouth opening in the vertical plane not directed toward physical material, not occurring during a period of struggling, and not coincident with tongue protrusion
overt chewing movements: distinguished from subtle chewing movements by the visibility of the interior oral cavity
subtle chewing movements: distinguished from overt chewing movements in that the interior oral cavity is not visible
References
Tomiyama K, McNamara FN, Clifford JJ, Kinsella A, Koshikawa N, Waddington JL. Topographical assessment and pharmacological characterization of orofacial movements in mice: dopamine D(1)-like vs. D(2)-like receptor regulation. Eur J Pharmacol. 2001 Apr 20;418(1-2):47-54.
PubMed 11334864