Wahlsten1 project protocol
Survey of motor activity, behavior, and forebrain morphometry in 21 inbred strains of mice across two laboratories (2003)
Wahlsten D, Crabbe JCWith: Bachmanov A, Finn DA, Cooper S, Bryden C, Metten P, Sibert J, Moisan B
Introduction: Comparative studies of behaviors and phenotypic characteristics on a wide range of inbred mice are based on the caveat that genetics play a major role, and that environmental influence may be significant, but controllable. To illustrate this, simultaneous studies of identical behavioral tests given under standardized conditions to identical strains in two different laboratories are expected to generate comparable results. Therefore, any difference in results, however subtle, implicates the inevitable variations in the research facility's environment. On the other hand, apparent robustness and replicability of the behavioral test results or phenotypic characteristics validate the genetic differences between inbred strains of mice.
Project protocol — Contents
Overall workflow
Equipment common to multiple procedures
Reagents, supplies, and solutions
Notes for all procedures in this project
Definitions & formulas
Procedure: Open Field Test (OFT)
Procedure: Elevated Plus Maze (EPM)
Procedure: Forebrain morphometry
References
Each mouse was tested for all procedures. Mice were not pretreated with drugs, special diet, or exposed to other environmental test conditions.
Day Test #Trial Duration Open Field Test Elevated Plus Maze Forebrain morphometry Equipment common to multiple procedures
- A close circuit TV (CCTV) camera is situated directly above the center of the apparatus for video-recording behavioral performance during testing.
- Computerized tracking system.
- A TV monitor for real-time observation of mice behavior.
- Transport rack, testing/transport/holding cages.
Reagents, supplies, and solutions
The apparatus is cleaned with tissue (Kimberly-Clark Kim-Wipes EXL #34256) moistened with Small Animal-S.O.X. (Hagen Inc. #H-30), and then allowed to dry completely.
Notes for all procedures in this project
- In any given session, all the mice are transported in their home cage(s) to (and from) the testing room at once on a small rack, where they are kept and acclimated for at least 30 min. up to 1 hour before testing begins.
- A mouse about to be tested is transiently transferred into a clean testing cage, that is placed on a piece of foam pad over a small table next to the rack, and that is further kept 1 to 1.5 m distance away from the test apparatus.
- As the transport (testing) cage is carried to the cubicle or maze and is held at approximately the same height as the apparatus, the mouse about to be tested is gently grasp by the tail using gloved fingers and placed in the center of the test apparatus facing distally. This process is done gently, smoothly, and continuously to facilitate testing and avoid extraneous disruptions.
- Distressing noises from any source is checked or avoided.
- Each testing session is composed of 5 x 1-minute trials.
- At the completion of each trial, the mouse is gently picked up with a pair of forceps and returned to the testing cage nearby. As the testing cage is brought back to the rack in the holding area, a mouse that has finished its session is returned into its home cage.
- Fecal boli are counted and recorded, as well as any noteworthy observations, such the quality of tracking.
Average speed is calculated as = (sum of the distances traveled (in cm) after 5 trials) ÷ (total duration for all 5 trials or 5 minutes (= 300 sec.).
Average distance traveled is calculated as = (total distances traveled (in cm) after 5 minutes) ÷ (number of trials (5)).
-------------------------------
Purpose: To measure strain differences in locomotor activity, anxiety and exploratory behavior using the open field test, and assess the stability of behavioral differences among the strains in two laboratories.
Equipment
- The open field apparatus is a 40 x 40 x 30 cm high clear acrylic box with no top or bottom. It is situated within a 50 x 50 x 60 cm testing cubicle made at the U. of Alberta (Wahlsten D et. al., 2006).
- A clean sheet of 18.5" x 19.5" paper cut from a roll of rose-colored butcher's wrapping paper is placed under the open field (waxed side up) before each trial begins.
Environmental test conditions: Lighting is maintained dimly at 30 Lux by using opaque white filters that covered all 4 fluorescent light bulbs attached to the cubicle lid.
Procedure: Open Field Test (OFT)
- While the computer tracking system is being set-up, a background snapshot is taken, and then a mouse is readied and held conveniently within the testing cage at the level of the cubicle.
- Once the mouse is properly positioned and testing cubicle door is closed the trial begins.
- The open field chamber is prepared for the next mouse by discarding and replacing the disposable floor paper, and the lower inside surface is cleaned.
----------------------------------------
Purpose: To measure strain differences in anxiety and exploratory behavior using the elevated plus maze, and to assess the replicability of observed strains differences in two laboratories.
Equipment
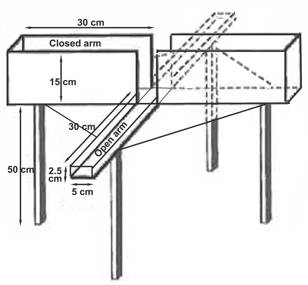
- The elevated plus maze apparatus is constructed in the Department of Psychology shop in Edmonton and consisted of 4 arms 5-cm x 30-cm that converged at a center square 5-cm x 5-cm zone, and is mounted on a clear plastic pedestal that elevated the 4 arms 50 cm above the floor (Wahlsten D et. al., 2006).
- Of the 4 arms two have 15 cm high, clear plastic walls ("closed" arms) and two have a small rim of clear plastic 2.5 mm high ("open" arms). See above figure for details. The walls of each arm are easily removed for cleaning in between mice.
Procedure: Elevated Plus Maze (EPM)
- Once the computer and monitoring system is primed, the experimenter is poised at the end of one open arm to position the mouse with its head and forelimbs in the center square and its hind limbs and tail in the open arm closest to the experimenter.
- The progress of the tracking system on the computer screen is followed behind one of the lights about 1 to 1.5 m from the maze, where potential shadows are not shed nor easily visualized by the mouse.
- Time between trial pause and resume, in the event a mouse falls or jumps off an open arm, is not included in trial duration.
- At the end of the trial, to facilitate the retrieval of a mouse in a closed arm, the high wall is gently and quietly removed.
- For cleaning, the rest of the walls from all four arms, including the "open" arms, are removed. The maze is disassembled and allowed to dry, while the computer and the next mouse to be tested is prepared for the next trial.
Environmental test conditions: The maze is located in a room where the surrounding visual cues are absent or at least comparable or symmetrical with regard to open versus closed arms. Lighting is provided by a frosted tungsten light bulb that is suspended at each corner of the frame with no more than 100 Lux.
----------------------------------------
Purpose: To investigate the replicability of brain morphometric measurements, such as brain weights, corpus callosum, hippocampus and anterior commisures sizes, across inbred strains of mice.
Abbreviations, comments, and measurements
Brain measurement workflow
Each mouse was previously tested for all behavioral procedures.
Equipment
- Micro-dissecting tools: forceps, curve and straight iris scissors, scalpel and blades.
- Triple beam balance
- Microscope/dissecting scope, equipped with image capture capability.
Procedure: Forebrain morphometry
I. Fixative and staining solutions preparation
* 2.5% sodium thiosulfate in distilled water
- * Physiological saline
- 0.9% sodium chloride in distilled water; cool to 4C
- * 10% neutral buffered formalin
- for 1 L: to 900 mL distilled water, add
- 4.0 g sodium phosphate monobasic
- 6.5 g sodium phosphate dibasic (anhydrous)
- 100 mL 40% formaldehyde solution (containing methanol as stabilizer)
- * 0.2% buffered gold chloride stain (Schmued, 1990; Wahlsten et al., 2003)
- for 1 L: to 1000 mL distilled water, add
- 1.8 g gold chloride [HAuCl4 . 3H2O] Fisher Scientific, #G54-5
- 0.33 g NaH2PO4 . H2O
- 3.6 g Na2HPO4
- 9.0 g NaCl
- [this stain can be reused]
II. Mouse euthanasia (consult veterinarian)
a. CO2 gas applied at full concentration in chamber until all breathing stops for at least 1 min.
III. Brain dissection and collection
a. Before dissection, the mouse is weighed to an accuracy of 0.1 g.
b. Using sturdy dissecting scissors, cut the scalp up the middle from the nape of the neck to the nose. Then remove the muscle and membranous tissue from the posterior part of the skull over the cerebellum, using scissors and scraping with a scalpel blade. Note that this method does not require removing the head from the torso.
c. Using small but sturdy scissors, cut the skull on both sides from the foramen magnum to the olfactory bulbs and remove the flap of skull. This procedure can also be done with a pair of curved, pointed, toothless Dumont forceps. Be sure to keep the tips of the scissors or forceps away from the midline of the cerebellum and cerebral cortex.
d. Gently pry the brain from the skull, cut the trigeminal and optic nerves, and let the brain fall into a bottle of fixative at room temperature (at least 20 mL per brain); seal bottle and label with an assigned code so that "blind" measurements may be taken without revealing the identity of the animal.IV. Brain fixation in 10% formalin
Agitate gently once or twice within the first hour so that the brain changes position and the solution mixes well. Keep the bottle sealed tightly for at least 1 wk.
V. Fixed brain trimmed according to standard configuration
a. Remove the brain from the fixative with a spatula and trim away parts that are often damaged during extraction, including (i) the paraflocculi of the cerebellum, (ii) the optic nerves up to the optic chiasm, (iii) the olfactory bulbs in front of the cortex. Cut the spinal cord at the base of the medulla oblongata.
b. Blot the brain gently by rolling it on a non-porous plastic surface or counter top until no further drops of solution remain on the surface.VI. Obtaining fixed, trimmed brain weights
a. Place the brain in a plastic or metal weighing dish on the balance. Calculate weight to an accuracy of 1 mg.
b. Return brain to the fixative in the bottle.a. Remove the brain from the fixative and place on a smooth surface, dorsal side up. The surface should have a straight edge guide for aligning the midline of the brain.
b. Use a fine razor blade or disposable microtome blade with a long, straight edge to cut the brain in half, starting at the longitudinal cerebral fissure and using one continuous motion from top to bottom. A blocking jig with guides perpendicular to the surface may aid in cutting. When the cut is correct, the base of the brain will be separated into two mirror-image parts. Then return the two halves to the fixative.VIII. Staining brain using gold chloride solution
a. Half of the brain is rinsed in physiological saline at 24°C for a few seconds.
b. Rinsed brain is stained with the gold chloride in an incubator at 37°C for 1-2 h to reveal myelinated axons. Monitor the progress of the staining every 15 min and stop when the commissures appear clearly and have a brown or brownish-purple color. The stain will work at room temperature but requires more incubation time. Note that gold chloride stain can be reused until exhausted.
c. Stained brain is placed in fresh 2.5% sodium thiosulfate solution for about 5 min to stop the staining reaction and preserve the gold chloride stain.
d. Stained half brain is returned back in the fixative solution with the unstained half.IX. Midsagital plane of stained half is viewed, digitally captured, and measured at the commisures.
a. Remove the stained half and place the flat surface against a microscope slide coverslip so that it adheres firmly by surface tension.
b. Invert the slide and support it with a 3.5 cm disposable petri dish so the brain does not touch the microscope stage. Adjust illumination to obtain a clear image through a stereoscope.
c. Capture the image with a video imaging system and save the image to a disk. Return stained half brain to fixative solution in the event measurements must be repeated.
d. Use an image analysis program such as ImageJ (NIH) to make measurements. Define an area of interest and use contrast enhancement to improve the image. Draw an outline of each commissure with the computer's mouse and find the area, and then draw a straight line between the two furthest points to find maximum length. When in doubt about the edge where the line should be drawn, look though the microscope.
Investigators Notes: Because certain fine distinctions must be made, it is advised to have the original stained brain available for reference via the stereoscope. When in doubt about the proper boundary of a structure, refer to the stereoscope image, which serves as the final authority. Be sure to make notes on any doubtful borders that may need to be rechecked. It is also helpful to refer to an authoritative atlas of the mouse brain; we use Sidman, Angevine and Taber Pierce (1971). The observer must be able to identify several structures that are commonly present at or near the midsagital plane. It may be helpful to examine some stained microscope slides in order to become familiar with the appearance of these structures.
References
Bishop KM, Wahlsten D. Sex and species differences in mouse and rat forebrain commissures depend on the method of adjusting for brain size. Brain Res. 1999 Jan 9;815(2):358-66.
PubMed 9878827 Crabbe JC, Wahlsten D, Dudek BC. Genetics of mouse behavior: interactions with laboratory environment. Science. 1999 Jun 4;284(5420):1670-2.
PubMed 10356397 Franklin KBJ, Paxinos G. The Mouse Brain in Stereotaxic Coordinates. (book) Academic Press, 1997.
Schmued LC. A rapid, sensitive histochemical stain for myelin in frozen brain sections. J Histochem Cytochem. 1990 May;38(5):717-20.
PubMed 1692056 Sidman RL, Angevine JB, Taber Pierce E. Atlas of the Mouse Brain and Spinal Cord. (book) Harvard University Press, 1971.
Wahlsten D, Colbourne F, Pleus R. A robust, efficient and flexible method for staining myelinated axons in blocks of brain tissue. J Neurosci Methods. 2003 Mar 15;123(2):207-14.
PubMed 12606069 Wahlsten D, Hudspeth WJ, Bernhardt K. Implications of genetic variation in mouse brain structure for electrode placement by stereotaxic surgery. J Comp Neurol. 1975 Aug 15;162(4):519-31.
PubMed 1097481 Wahlsten D, Metten P, Phillips TJ, Boehm SL 2nd, Burkhart-Kasch S, Dorow J, Doerksen S, Downing C, Fogarty J, Rodd-Henricks K, Hen R, McKinnon CS, Merrill CM, Nolte C, Schalomon M, Schlumbohm JP, Sibert JR, Wenger CD, Dudek BC, Crabbe JC. Different data from different labs: lessons from studies of gene-environment interaction. J Neurobiol. 2003 Jan;54(1):283-311.
PubMed 12486710