Taft1 project protocol
Performance survey following assisted reproductive technologies in females of 10 inbred strains of mice (2006)
Taft RA, Byers SLWith: Payson SJ
Project protocol — Contents
Equipment
Common reagents, supplies, solutions
Procedure: superovulation
Procedure: in vitro fertilization
Procedure: embryo freezing and thawing
Procedure: embryo transfer
Data
References
Definitions
COCs: cumulus oocytes complexes are obtained following superovulation, and are the primary source of mature oocytes for in vitro fertilization (IVF).
eCG/PMSG: equine chorionic gonadotrophin/pregnant mare serum gonadotrophin, originates in the endometrial cups and detected in the blood during pregnancy. This steroid hormone is used in promoting or stimulating the growth of follicles, and used in combination with prostaglandins to promote superovulation.
hCG: human chorionic gonadotrophin, is a steroid hormone with characteristics like those of luteinizing hormone (LH). It is produced by the ovaries and secreted in the placenta during gestation, and is used to induce ovulation and for synchronization of estrus.
HTF: is a synthetic human tubal fluid media, and is used for in vitro fertilization (IVF), and embryo transfer (ET).
M2: is a chemically synthesized embryo culture media, and is used in combination with cryoprotectant propylene glycol (PG) in the freezing of 2-cell embryos.
Workflow, sampling, and experimental treatment*
*(Byers & Taft, 2005)
Day Manipulation Hormone Rx Media/Tx Females are injected i.p. between 5-8 pm Females are induced to ovulate mature oocytes Oviducts are harvested Caudal epididymis and vas deferens are harvested Female recipients in proestrus are mated to vasectomized males Pooled COCs are added with 5 µL sperm Count 1-cell, 2-cell embryos, dead, fragmented oocytes Two-cell embryos are pooled Females with vaginal plugs are used as recipients Uterus is harvested: count live/dead fetuses and implantation sites
- Bench-top incubator (Barnstead International, IA, USA) with mixed gases (5%CO2, 5%O2, balanced with N2), with constant temperature (37°C), and pH (7.2-7.4)
- Modular incubator chamber (Billups-Rothenberg Inc., Del Mar, CA, USA)
- Dissecting microscope, 37°C tissue-culture incubator, micro-dissecting instruments.
Common reagents, supplies, solutions
• Human tubal fluid (HTF) medium is prepared in advance (Sigma, St. Louis, MO, USA)
Synthetic human tubal fluid (HTF) medium recipe:
Ingredients Components Sodium chloride (NaCl) Potassium chloride (KCl) Magnesium sulfate (MgSO4⋅7H20) Potassium phosphate (KH2PO4) Calcium chloride (CaCl2⋅2H2O) Sodium bicarbonate (NaHCO3) Sugar (energy source) Glucose Sodium pyruvate Sodium lactate Penicillin Streptomycin Phenol red (1%) dye BSA (bovine serum albumin)
- Light mineral oil (Fisher Scientific, USA) is washed with HTF.
- The HTF and oil are allowed to stand until the oil is separated and easily decanted, and then filtered (0.8 mm filter) before use.
- The used HTF wash is discarded.
Common to all procedures
Refer to Byers & Taft, 2005 for precise details of this study.
a. Oocyte donor females are 17-27 days of age and sperm donor males are 8-31 weeks of age.
b. Hormonal injections are delivered intraperitoneally (i.p.) between 5-8 pm.
c. Males and females (13-14.5 hours post-hormonal treatment) are euthanized by cervical dislocation.
d. Female CByB6F1/J mice, 9 – 13 weeks of age and in proestrus, are mated to vasectomized CByB6F1/J males, to induce pseudopregnancy, and to serve as recipients.
e. Culture dishes and medium are equilibrated in 37°C tissue-culture incubator 30 min. before use.
f. Microdissecting instruments are autoclaved and sterilized before use.Environmental test conditions
Date of experiment is accomplished from September through October.
Harvested oocytes are scored as follows:
Oocyte/embryo score Description Normal or Live 2-cell 2-cell with intact zonae pelucida, homogenous cytoplasm, and morphologically sound Normal or Live 1-cell 1-cell with intact zonae pelucida, homogenous cytoplasm, and morphologically sound Dead Appear dark with granular cytoplasm Fragmented Show signs of blebbing or cellular swelling, resulting in breaks and fragmentation -----------
Purpose: To establish baseline performance values for 10 inbred strains following hormonal treatment to superovulate.
Reagents, supplies, solutions
- Equine chorionic gonadotrophin: eCG (PMSG; CalBioChem LaJolla, CA, USA)
- Human chorionic gonadotrophin hCG (Sigma, CG-10 St. Louis, MO, USA)
- Tissue culture dishes
Procedure: superovulation
Inbred strains of mice, listed as high priority on the Mouse Phenome Database, are used for their popularity as progenitors for recombinant inbred, consomic, or congenic strains, and in the creation of genetically modified animals. All strains (129S1/SvImJ, A/J, BALB/cJ, BALB/cByJ, C3H/HeJ, C57BL/6J, DBA/2J FVB/NJ, NOD/LtJ, and SJL/J) are subjected to the same procedures, thus allowing comparisons to be made among strains on the number of eggs produced in response to superovulation, the proportion of two-cell embryos produced following in vitro fertilization (IVF), and the proportion of live pups born following the transfer of fresh and thawed two-cell embryo.
a. On day 1, between 5-8 pm, donor females are first treated with 5.0 IU of eCG (or PMSG, pregnant mare serum gonadotrophin) to induce follicular development.
b. 46-50 hours later, on day 3, donor females are given 5.0 IU of hCG (human chorionic gonadotrophin) to induce ovulation of mature oocytes. Within approximately 12 hours post-hCG administration, ovulation occurs.
c. Following ovulation, 13-14.5 hours after the last hormone treatment, the oviducts are harvested, and placed in culture dish with 300 µL drop of fresh, 37°C, human tubal fluid (HTF) medium.
d. Using a dissecting scope and microdissecting forceps, the mass of oocyte/cumulus complexes (COCs) are released from the ampulla of the oviducts.
e. The cumulus oocyte complexes are pooled so that 2-6 COCs (more for A/Js) are added with each fertilization drop containing sperms (see IVF or Byers & Taft, 2005).Definitions & formulas
Percent females responding to superovulation = (# females responding to superovulation ÷ # females injected to superovulate)*100.
Total oocytes per donor = (normal live oocytes per donor + dead oocytes per donor + fragmented oocytes per donor).
----------
Purpose: To establish baseline performance values for 10 inbred strains following in vitro fertilization of fresh or frozen embryos.
Reagents, supplies, solutions
- IVF culture dishes (60 mm x 15 mm standard BD Falcon petri dish (Fisher Scientific, USA))
- Sperm collection dishes (35 mm x 10 mm standard BD Falcon petri dish (Fisher Scientific, USA))
Procedure: in vitro fertilization
a. IVF tissue culture dish is prepared with the addition of one 250µL microdrop fertilization medium (HTF), two 150 µL microdrops of wash medium (HTF), and one 150 µL microdrop of culture medium (HTF), all covered light mineral oil.
b. Sperm collection culture dish is prepared with a 1mL microdrop of HTF covered with a layer of light mineral oil.
c. Both caudal epididymides and vas deferens are harvested and placed in the sperm collection dish prepared above.
d. The epididymides are sectioned several times and then incubated for 10 min to facilitate spermatozoa motility and collection. Sperm motility is visually assessed.
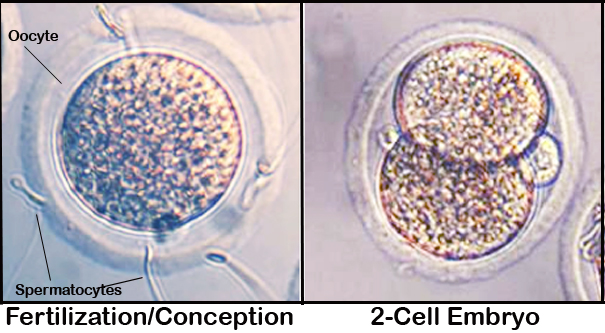
e. A 5 µL aliquot of epididymal sperm is placed in an IVF culture dish using a wide-bore pipette tip followed by the addition of pooled cumulus oocyte complexes (see superovulation or Byers & Taft, 2005), and then incubated for 4 hours in a modular incubator chamber at 37°C in 5% CO2, 5% O2, 90% N2. During this fertilization stage, the cumulus cells are displaced by the sperm; thus, facilitating the counting of normal, fragmented, and dead oocytes.
f. Oocytes appearing healthy are washed twice with two 150 µL microdrops of HTF to remove cellular debris (unbound sperms and cumulus cells) and then incubated overnight in a third 150 µL microdrops HTF culture medium, before the end of day 3.
g. On day 4, about 18 hours after the last wash, two-cell embryos, one-cell embryos, dead, and fragmented oocytes are assessed and counted (see Oocyte count).Investigators notes: Two-cell embryos are pooled, so that around 35-65 embryos per IVF dish are immediately transferred into pseudopregnant females as described in Embryo transfer or Byers & Taft, 2005.
Excess embryos are frozen or cryopreserved as described in Embryo freezing and thawing or Byers & Taft, 2005.
---------
Purpose: To establish baseline performance values for 10 inbred strains following embryo cryopreservation and embryo thawing.
Equipment
- Controlled rate freezer (Biotronics DB1, ZMS Inc., FL, USA)
- Liquid nitrogen and liquid nitrogen container (Thermo Forma, OH, USA)
- Pulled glass pipettes
- Water bath, temperature controlled
Reagents, supplies, solutions
- 1.0 M sucrose, 1.5 M propylene glycol (PG)
- 0.25-mL plastic insemination straws (IMV, Maple Grove, MN, USA)
- Critoseal (Fisher Scientific, USA)
- M2 medium (Glenister & Rall, 1999)
Procedure: embryo freezing and thawing
a. To freeze two-cell embryos, the embryos are first pipetted from the HTF medium, next put into M2 medium, and then finally a cryoprotectant solution of M2 with PG for 15 min.
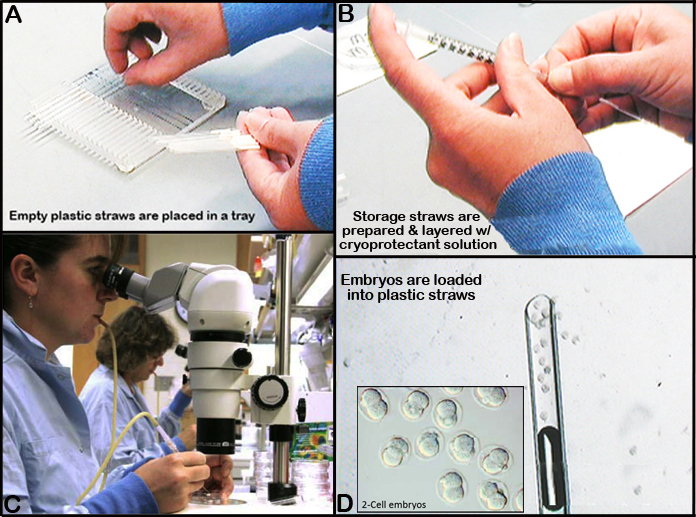
b. Storage straws are prepared and layered with 3.5 cm layer of sucrose medium, 0.5 cm air, followed by 0.7 cm of propylene glycol, and then 2.0 cm of air (See Figures 1 and 2 below).
c. Using a finely tapered pulled glass pipette, about 30 embryos are loaded in the propylene glycol layer of the prepared storage straw above before it is sealed with Critoseal.
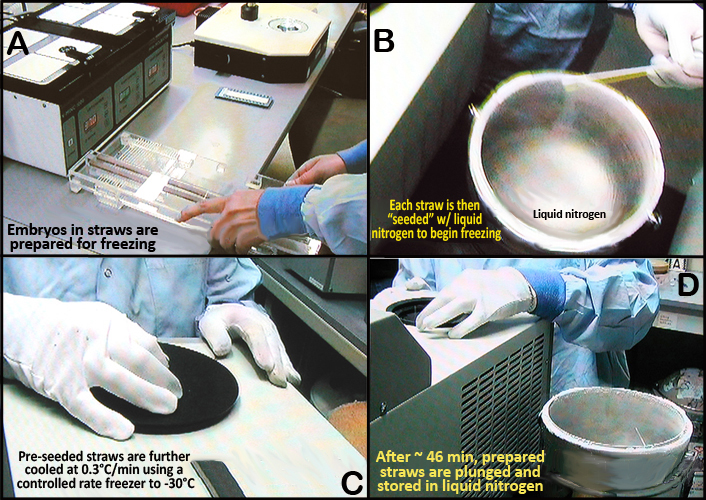
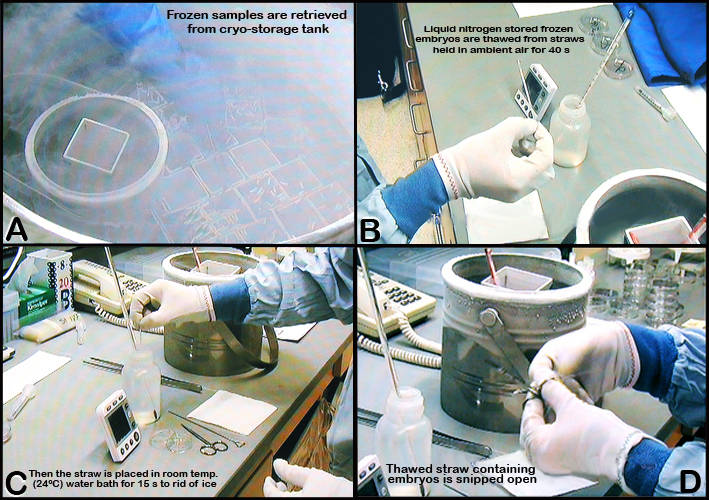
d. Embryos in straws are held at -7°C for at least 5 min before being seeded, cooled at 0.3°C /min using a controlled rate freezer to -30°C, and then plunged and stored in liquid nitrogen.
e. Liquid nitrogen stored frozen embryos are thawed from straws held in ambient air for 40 s, and then quickly placed in room temperature water bath for 15 s to rid of ice (See Figures 4 and 5 below).
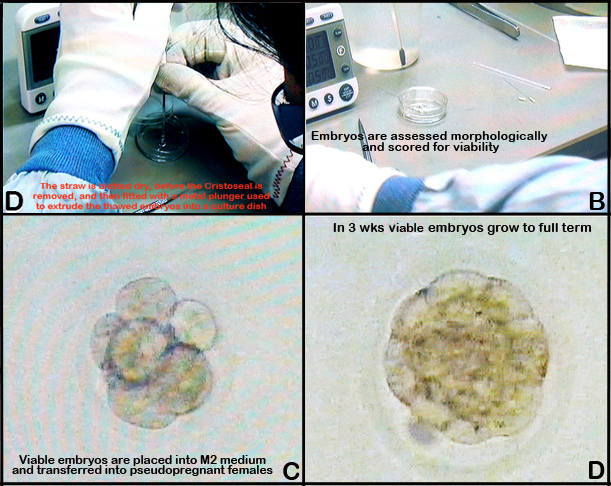
f. The straw is blotted dry, before the Cristoseal is removed, and then fitted with a metal plunger used to extrude the thawed embryos into a culture dish (See Figure 5 below).
g. The embryos are assessed morphologically and scored for viability (see Oocyte count); after 5 min they are placed into M2 medium and transferred into pseudopregnant females as described in Embryo transfer.Investigator notes: The number of embryos frozen for each strain varied depending on the number of embryos produced during the IVF procedure. For some strains, very few live embryos were recovered after thawing and this limited the number of embryo transfers performed for those strains (i.e., A/J, BALB/cJ, and SJL/J).
Figure 1.
Figure 2.
Figure 3.
Figure 4.
Figure 5.
------------
Purpose: To establish baseline performance values for 10 inbred strains following the transfer of fresh or previously frozen embryos into pseudopregnant recipient females.
Procedure: embryo transfer
a. Pseudopregnant recipient females are mated (day 3) and then examined the following morning (day 4) for vaginal plugs.
b. Only females with vaginal plugs are used as recipients for the transfer of 7-13 fresh or previously frozen (see embryo freezing and thawing) embryos into one oviduct.
c. Embryo transfer is accomplished in three separate days.
d. Pregnant embryo recipients are fed an 11% fat rodent chow (LabDiet1 5LA6, St. Louis, MO, USA) throughout gestation.
e. By day 17-18 of pregnancy, or 19-21 days after fertilization, the recipient female uterus is harvested and the numbers of live neonate, implantation sites, and resorbed fetuses are recorded:
Score Description Live neonate Neonates are able to move and breathe, and have a sanguine color Dead Fetuses appear morphologically normal, but never moved or breathed Implant sites Sites are seen as small dark spots on the uterine wall in the absence of an embryo Figure 6.
Data collected by investigator
- Superovulation: total oocytes per donor
- In vitro fertilization: number of 2-cell and 1-cell embryos, dead embryos, and fragmented oocytes
- Fresh embryo transfer: number of live pups, implant sites, and resorbed fetuses
- Frozen embryo transfer: number of live pups, implant sites, and resorbed fetuses
- Note: Because of the nature of these data, individual data set rows (and the N associated with each strain mean) can represent either culture dishes, donors, or embryos depending on context. No animal ids or similar case identifiers are available.
References
Sztein JM, Noble K, Farley JS, Mobraaten LE. Comparison of permeating and nonpermeating cryoprotectants for mouse sperm cryopreservation. Cryobiology. 2001 Feb;42(1):28-39.
PubMed 11336487