Puk1 project protocol
Survey of eye size parameters using optical low coherence interferometry in 4 inbred strains of mice (2006)
Puk O, Graw JWith: Dalke C, Favor J, and Hrabě de Angelis M
Project protocol — Contents
Workflow and sampling
Equipment
Reagents, supplies, and solutions
Procedure
Data
References
Each mouse anesthetized for in vivo biometry Anesthetized mouse positioned in an adjustable platform for measurement Pupillary axis aligned with the measurement axis via LED signals Series of longitudinal scans conducted Eyes treated to prevent dehydration Mice allowed to recover Interferograms analyzed and eye parameters calculated using dedicated software * 13 wk data were accessioned as MPD measurements and are eligible for analysis with MPD tools.
Data for all ages are available (Puk1).
• ACMaster for in vivo biometry using low coherence interferometry for the mouse eye
Measurement principle (for more details see Schmucker et. al., 2004)
"Biometrical data of the mouse eye are collected with a device based on optical low coherence interferometry (OLCI), the Carl Zeiss "ACMaster". The principle of OLCI is based on a Michelson interferometer. The light source is a low coherence superluminescent laser diode (SLD) that emits an infrared light with a peak emission at 850 nm and a half-band width of 10 nm. Due to the broadened bandwidth, the coherence length is rather short (about 10 µm), compared to standard laser diodes, in which it is about 160 µm. Output energy is 450 µW. The infrared laser beam emerging from the LED is divided into two perpendicular beams by a semi-silvered mirror. One part is transmitted through the semi-silvered mirror and reaches a stationary mirror. The other part is reflected and reaches a mirror that can be moved along the light path with high positional precision. After reflection from both mirrors, two coaxial beams of about 50 µm diameter propagate to the eye, where they are reflected off from the cornea, the lens and the retinal pigment epithelium (RPE) close to the vitreo-retinal interface. Interference between both beams can only occur when their optical path lengths are matched within the coherence length. The occurrence of interference is detected by a photo cell and recorded as a function of the displacement of the movable mirror. Due to the usage of coaxial beams, the measurements are largely insensitive against longitudinal eye movements. The scanning time of the movable mirror is about 0.3 s. The resolution of the system is limited both by the coherence length, which is inversely proportional to the bandwidth of the SLD, and by the precision by which the position of the movable mirror can be controlled. In the human eye, a measurement precision in the range of 2 µm has been described in corneal thickness measurements and of 5-10 µm for the anterior chamber depth and lens thickness measurements.
The major reflections in the eye occur at the anterior corneal surface and at the RPE. Accordingly, the interference signals are most conspicuous at these two layers. The software of the "ACMaster" is designed to measure the anterior segment in human eyes. This means that it expects to find reflecting surfaces at about 0.5 mm behind the anterior corneal surface (which would correspond to the thickness of the human cornea) and a second major reflection between 2 and 5 mm distance (the anterior surface of the lens). The software could be used to measure mouse eyes because reflecting surfaces are present within the ranges accepted by the software: the distance to the anterior surface of the lens of the mouse eye is in the range of the thickness of the human cornea, and the distance to the RPE in the back of the eye is in the range of the distance of the anterior lens surface in human eyes. This means that the software had to detect anterior corneal surface, anterior lens surface, and the RPE to provide biometric data. Anterior chamber depth, is defined as the distance from the anterior corneal surface to the anterior surface of the lens. The peak of the posterior lens surface is detected only in a few measurements. Therefore no consistent data on lens thickness are provided."
• Zeiss Meditec modified software optimized for the dimensions of the mouse eye
Figure 1. ACMaster interferometer
Reagents, supplies, and solutions
• eye ointment (Regepithel, Alcon Pharma, Freiburg, Germany) to avoid eye damage from dehydration
•137 mg ketamine and 6.6 mg xylazine per kilogram body weight (or 0.1â0.2 mL of a mixture of 1.2 mL 10% ketamine hydrochloride and 0.8 mL 2% xylazine hydrochloride, dissolved in 8.0 mL sterile physiological (~0.9%) saline)
• needles and syringes
• 95% ethanol for cleaning and disinfectant
Procedure for optical biometry
In vivo measurement procedure (for more details see Schmucker et. al., 2004)
The refractive power of the eye depends on three factors: the strength of the cornea, the strength of the lens, and the length of the eye. It is possible to calculate the strength of the lens that will give the best refraction, if both the strength of the cornea and the (axial) length of the eye are known. Hence, biometry is the process of measuring the strength of the cornea (keratometry) and the length of the eye, and using these data to determine the perfect intraocular strength of the lens.a. All measurements are performed the same time between 10 a.m. and 4 p.m.
b. Before conducting biometric measurements, mice are anesthetized with an intraperitoneal (i.p.) injection of ketamine hydrochloride and xylazine hydrochloride cocktail.
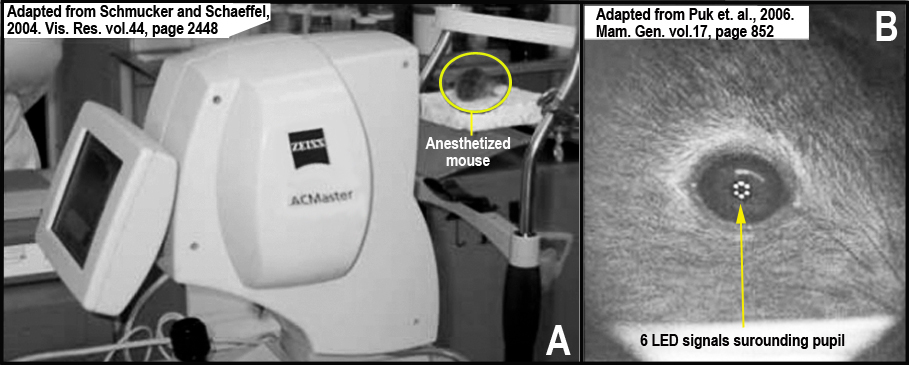
c. Subsequently, the animals are positioned on an adjustable platform that is screwed to the chin rest of the device (see Figure 2 A below).
d. The pupillary axis of the eye is aligned with the measurement axis, and the distance of the eye to the measurement head is adjusted to approximately 70 mm, using 6 infrared LED signals arranged in a circle and visible on the cornea when focused under high magnification (see Figure 2 B below).
e. Then, a series of approximately 20 longitudinal scans are performed within a few seconds.
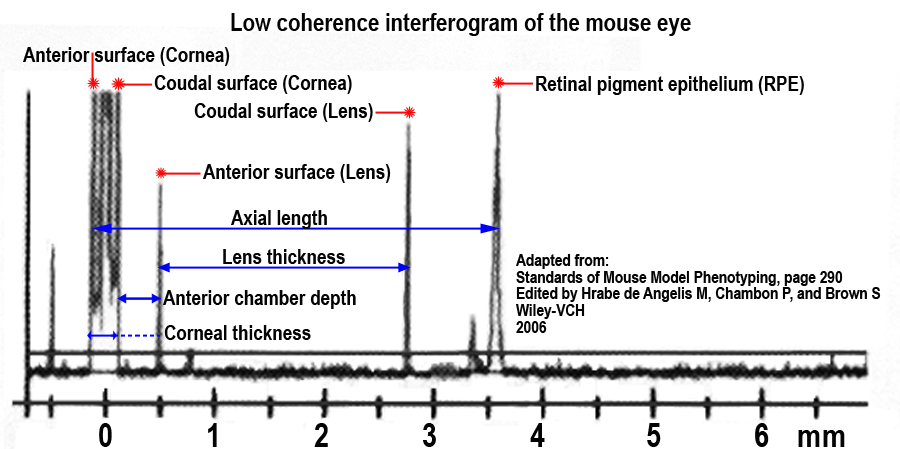
f. Only those scans which showed clear peaks at the cornea, the anterior lens surface and the retino-vitreal interface are used for deriving the various measurements including eye length, corneal thickness and anterior chamber depth in each eye (see Figure 3 below).
g. After each series of measurements, eyes are treated with ointment to prevent damage from dehydration.h. Modified software optimized for the dimensions of the mouse eye is used to enable peak recognition automatically (Zeiss Meditec; posterior cornea signal = 70-150 µm, anterior lens signal = 300-700 µm, posterior lens signal = 2100-2900 µm, retina = 3000-3900 µm, see Figure 3 below).
i. For each mouse, the left and the right eyes are measured independently; the value for each eye is the mean of 15-60 individual measurements in an interval of 30 s to 1 min; this individual mean is used for all subsequent calculations.
j. At least 5 mice of each sex of each strain are measured in each age group.
k. Mean values and standard deviations of axial length, lens thickness, and anterior chamber depth are calculated at 4, 6, 8, 11, 13, and 15 wks.
l. Microsoft Excel is used for analysis.
Figure 2 A. Another view of the ACMaster interferometer. Note the adjustable platform designed to hold an anesthetized mouse (yellow circle) during measurement. Figure 2 B. A high magnification of the eye and the cornea with infrared LED signals arranged in a circle (yellow arrow).
Figure 3. Schematic illustration of a low coherence interferogram of the mouse eye, where the intensity of the peak is plotted against the optical path length.Definitions and formulas
LED: light emitting diode, is an electronic light source.
CT: Corneal thickness
AD: Aqueous depth (back of cornea to front of lens)
LT: Lens thickness
AL: Eye length (front of cornea to retinal peak)
Figure 4 A. Schematic illustration of the different parts of the eye and various parameters (marked by red dot and arrows) measured by the interferometer. Figure 4 B. Diagrammatic illustration of the parameters measured in the eye (CT= corneal thickness, AC= anterior chamber depth, LT= lens thickness, AL= axial length).
References
Astbury N, Ramamurthy B. How to avoid mistakes in biometry. Community Eye Health. 2006 Dec;19(60):70-1.
PubMed 17515971 FullTextFuchs H, Gailus-Durner V, Adler T, Pimentel JA, Becker L, Bolle I, Brielmeier M, Calzada-Wack J, Dalke C, Ehrhardt N, Fasnacht N, Ferwagner B, Frischmann U, Hans W, Hölter SM, Hölzlwimmer G, Horsch M, Javaheri A, Kallnik M, Kling E, Lengger C, Maier H,. The German Mouse Clinic: a platform for systemic phenotype analysis of mouse models. Curr Pharm Biotechnol. 2009 Feb;10(2):236-43.
PubMed 19199957 Hrabe de Angelis M, Chambon P, and Brown S, Editors. Standards of Mouse Model Phenotyping. (book) Wiley-VCH, 2006.
Schmucker C, Schaeffel F. In vivo biometry in the mouse eye with low coherence interferometry. Vision Res. 2004;44(21):2445-56.
PubMed 15358080