Jaxwest2:
Neoplasia induction in NOD.CB17-Prkdcscid/J female mice by passage of live tumor cells (HT29) (2007)
Jackson Laboratory
Yan Yang With: Yang Y, Rodriguez R, Braun M, Clary D Click on "Download" link or button below, to see the available downloads for this project.
| Institutional authorship |
Jackson Laboratory
|
| Investigators |
Yan Yang The Jackson Laboratory, Bar Harbor, ME |
| Participants | Yang Y, Rodriguez R, Braun M, Clary D |
| Contact | Yan Yang yan.yang@jax.org
|
| Affiliated Center | JAX In Vivo Services |
| One-strain study | |
| MPD identifiers | Jaxwest2 MPD:257 |
| No updates/corrections. Initial release date: 06/2007. | |
|
Click above to copy-paste the entire citation for this MPD web page. |
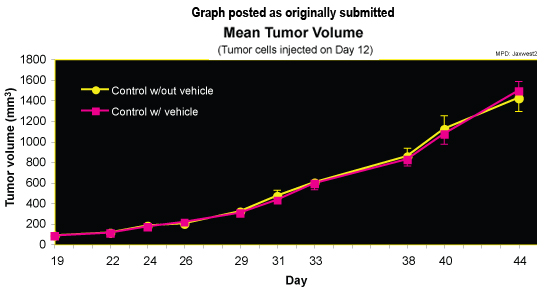
Purpose: To provide normative data for testing biologically-active compounds in immunodeficient female NOD.CB17-Prkdcscid/J strain. This strain is an excellent host for xenografts, including the human colorectal adenocarcinoma cell line, HT-29, used here for tumor induction.
By posting this data, JAX® In Vivo Services contributes to community research resources and fulfills an agreement with clients to make normative data public.
Available download: Jaxwest2.xls
X = Each of these measurements is replicated for test Day X (see protocol for more information).
The data set contains these columns: strain sex mouse_ID birth date of birth arrival date of arrival at test facility cage_code cage code at test facility HT29_inj tumor cells (HT29) injected exp_group experimental group: Control w/out vehicle; Control w/vehicle; Test Group (pre-treatment data only) bw_X body weight on Day X (after arrival) g bw_chg_X change in body weight from Day 12 to Day X % tumor_L_X tumor length on Day X mm tumor_W_X tumor width on Day X mm tumor_H_X tumor height on Day X mm tumor_vol_X tumor volume on Day X mm3 tumor_chg_X change in tumor volume from 1 wk after HT29 injection (Day 19) to Day X % tumor_wt_44 tumor weight at necropsy (Day 44) g survival_500 number of days after HT29 injection that tumor volume reached 500 mm3 n survival_1500 number of days after HT29 injection that tumor volume reached 1500 mm3 n