Chesler3 project protocol
Multi-system assessment of 8 inbred founder strains and 360+ emerging lines (pre-CC) of the Collaborative Cross (2011)
Chesler EJ, Philip VM, Voy BHWith: Sokoloff G, Ackert-Bicknell C, Striz M, Churchill GA, Lariviere WR, Palmer AA, OHara BF
Project protocol — Contents
Workflow and sampling
Equipment and supplies
Reagents and solutions
Procedure for reproductive performance
Procedure common to behavioral tests
Procedure for scoring behavioral wildness
Procedure for open field test (OFT)
Procedure for light-dark test
Procedure for sleep analysis
Procedure for conducting hot plate test
Procedure for conducting tail clip test
Procedure for conducting complete blood cell count (CBC)
Procedure for obtaining blood chemistry
Procedure for necropsy and morphometry
Definitions
Data
ReferencesWorkflow
Mice bred and maintained in breeding pairs Mice moved to holding room and housed for 3 wks Mice scored for wildness Mice assessed using an open field arena Mice evaluated in light/dark chambers Sleeping patterns examined for 5 days Mice sleep deprived and assessed for sleep pattern on third day Mice subjected to thermal nociception test Mice subjected to mechanical nociception test Mice bled via retroorbital sinus Blood collected for blood chemistry Mice fasted overnight and bled for fasting glucose level Mice fasted overnight before necropsy and dissected; hind limb disarticulated and preserved Hind limb dissected to isolate femoral bone
- Open field apparatus: white, square, opaque Plexiglas box (39 x 39 x 39 cm) with a red floor, illuminated evenly at 300±10 lux in a 9-foot x 15-foot room
- Noldus Ethovision XT tracking system (Noldus Information Technology)
- LightâDark box
- Hot plate (IITC Inc. Hotplate Analgesia Meter Model 39)
- Plexiglas-bound arena open at the front and measuring 13.5 in (L) x 16 in (W) x 15 in (H)
- Alligator clips
- Stopwatch
- Denim (restraining) pockets
- Blood collection kit
- Portable Handheld i-STAT (Abbott Laboratories, Abbott Park, IL)
- Piezoelectric grid and sleep chamber system
- Hot plate analgesia test meter for mice (IITC Inc. Hotplate Analgesia Meter Model 39) with a transparent Plexiglas cylinder (15 cm (D) x 22.5 cm (H)) and lid enclosure
- Scil-Vet ABC blood analyzer (Scil, Gurnee, IL)
- Bayer Glucometer Elite XL (Bayer AG)
- Dissecting kit
- Balance scale
- Anticoagulants: EDTA and lithium heparin
- Abbot i-STAT Chem 8 panel (Abbot) cartridge: Each test cartridge contains chemically sensitive biosensors on a silicon chip that are configured to perform specific tests. i-STAT single-use cartridge uses advanced biosensor technology that applies microfluidics to process the most comprehensive range of clinical tests in a single platform. This cartridge-based system obtains measures of sodium, potassium, chloride, ionized calcium), CO2, glucose, urea nitrogen (BUN), creatinine, hematocrit (HCT), hemoglobin (HGB), and anion gap.
- 95% Ethanol
- Disinfectant or cleaning solution MB-10 (QuipLabs)
Acclimation to test conditions
Mice are acclimated to the examination room for 1 h before testing.
Procedures for high-throughput phenotyping strategy and study design
The phenotyping strategy is designed to capture a panel of complex traits amenable to high-throughput analyses while broadly reflecting behavior, morphology, and physiology. Retired breeders from the cross population are phenotyped across generations.
I. Procedure for reproductive performance
a. Litters are weaned at ~3 wk of age into breeding pairs determined by the CCWORKS software-assisted mating system (Philip et al. 2012).
b. Dates of birth (DOB) for each breeder are recorded.
c. Each breeding pair is monitored regularly for signs of first kindling or imminent parturition.
d. Upon noting the day of parturition, the number of total pups are counted regardless of sex.
e. Parental age is obtained and the latency to first litter is calculated.
f. Mice are maintained in breeding pairs until they entered the phenotyping protocol.Procedure common to behavioral tests
a. Retired breeders are separated into individual pens for phenotyping after the birth of grand-progeny.
b. At this time mice are housed in an adjoining holding room throughout the testing period.
c. A standardized enrichment device in the form of an elbow-shaped PVC pipe is added to each cage 1 wk in advance of testing.
d. Mice intended to begin testing the following week remain in place on one side of one rack.
e. Female mice are checked to ensure that they are not pregnant at the time of testing.
f. All activity monitoring tests (open field, light/dark, and modified visual cliff) are administered on separate days.
g. The testing is carried out in a temperature-, noise-, and light-controlled room.
h. Mice are acclimated to the room for 1 h before testing, and the light intensity for each arenaâs four corners and center point is adjusted to 300±10 lux.
i. Each mouse is picked up by the tail and then placed gently into the center of the arena with its nose pointed east.
j. All activity is recorded by a video camera mounted above the open field arena.
k. Activities are scored in real time by body point tracking using Noldus Ethovision XT tracking system.
l. Each apparatus is wiped clean between subjects to avoid olfactory cueing influencing activity and behavioral performance.II. Procedure for scoring behavioral wildness
a. During separation for phenotyping, mice are scored on the behavioral wildness scale (Wahlsten2), for which scores are obtained for the mouseâs response to capture and holding by the technician.
b. The scores are combined to form a total wildness score.III. Procedure for open field test (OFT)
a. Each mouse is placed in the center of an open field arena for a 10-min trial.
b. Activity is recorded by a video camera mounted above the open field.
c. Data recorded are configured for the following parameters using Noldus Ethovision XT software: number of crossings into the center of the field, distance traveled, the time spent in the periphery (thigmotaxis), the time spent immobile.
d. At the end of each trial, the mouse is removed from the arena and frequency of defecation and urination is recorded.IV. Procedure for light-dark test
a. Using the same open field apparatus, a dark insert dividing the arena into light/dark compartments is installed to test for anxiety-related behavior.
b. The compartments are separated with a guillotine door that is closed during placement of mice in the arena.
c. Mice are placed in the light compartment and behavioral performance is videographed over a 10-min trial.
d. Data recorded are configured for the following parameters: latency to enter the dark compartment, percent time spent in light compartment, total lightâdark transitions, distance traveled in the light side.
e. At the end of each trial, the mouse is removed from the arena and frequency of defecation and urination is recorded.V. Procedure for sleep analysis
a. Each mouse is placed in its own chamber over a piezoelectric grid within a sleep chamber system for a 5-d sleep analysis (Philip et al. 2012).
b. Mice are given free access to food and water while in the chamber.
c. The room is maintained on a 12:12-h light:dark cycle.
d. Mice are placed in the chambers between 9 and 10 a.m. on Day 1 and are removed on Day 5 at the same time.
e. Food and water are checked daily.
f. The mice are minimally disturbed, except during the sleep deprivation test on Day 3.
g. On Day 3, mice are disturbed by changing bedding, taking away nestlets, and placing them in brown paper bags.
h. Recorded data are analyzed for activity onset, time of peak activity, sleep bout length, total sleep time, sleep bout length after sleep deprivation, peak activity after sleep deprivation, and activity onset after sleep deprivation.VI. Procedure for conducting hot plate test
a. Mice are allowed to habituate to the testing room for at least 30 min.
b. Mice are then placed on a pre-heated metal surface within a hot plate analgesia test meter and enclosed by a transparent Plexiglas cylinder and lid.
c. The hot plate is pre-heated and maintained at 54°C.
d. The latency to respond with a jump or hindpaw lick or shake/flutter is measured to the nearest 0.1 s using a stopwatch.
e. Two latencies are recorded per mouse with inter-trial interval (ITI) of 30 s and maximum trial duration of 30 s.
f. If no response occurs within 30 s, the mouse is removed from the hot plate.
Analgesia test meter for miceVII. Procedure for conducting tail clip test
a. Two days after hot plate testing, the tail-clip mechanical nociception test is performed.
b. Mice are allowed 30 min of habituation.
c. Each mouse is enclosed in a Plexiglas-bound arena with an opening at the front and lightly restrained in a denim pocket.
d. An alligator clip with a rubber cuff around each jaw, exerting ~600 g of force, is applied to the tail 1 cm from the base and vertically oriented with respect to the table.
e. The mouse is immediately removed from the holder, and the latency to lick, bite, or grab the clip or bring the head within 1 cm of the clip is measured with a stopwatch to the nearest 0.1 s, after which the clip is immediately removed.
f. Each mouse is tested only once with maximum trial duration of 60 s.
g. The tail clip is removed if no response occurs after 60 s.VIII. Procedure for conducting complete blood cell count (CBC)
a. Blood is drawn for phenotypic assays following completion of behavioral testing.
b. Samples for CBC are collected by retroorbital sinus puncture into tubes containing EDTA as an anticoagulant.
c. At least 12 µL plus instrument dead volume (~ 25 µL) of whole blood is aliquoted into a sample tube, gently mixed with the index finger to assure proper mixture of anticoagulant.
d. Before blood cells count are obtained, a mouse specific smart card with pre-programmed blood cell parameters is inserted into the Scil-Vet ABC blood analyzer (Scil) (see image below, left panel).
e. The sample bar is then pushed to aspirate 12 µL of blood sample automatically (see image below, middle panel) and begin the counting process.
f. After 90 s, results are automatically printed, including lymphocyte counts and percentages (LYM, LYM %), monocyte counts and percentages (MONO, MONO %), granulocyte count and percentages (GRA, GRA %), and eosinophil count and percentages (EOS, EOS %), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV), mean platelet volume (MPV), neutrophils, hematocrit (HCT), hemoglobin (HGB), platelet count (PLT), red blood cell count (RBC), and white blood cell count (WBC) (see image below, right panel).IX. Procedure for obtaining blood chemistry
a. Samples for analysis of blood chemistry are collected by retroorbital sinus puncture into tubes containing lithium heparin as an anticoagulant.
b. Whole blood chemistry measurements are obtained using the Abbot i-STAT Chem 8 panel cartridge.
c. Prior to running a test, the Handheld is customized appropriately according to manufacturer's instruction to run the i-STAT Chem 8+ cartridge for a series of preset quality control diagnostics, from monitoring the quality of the sample to validating the reagent.
d. 2 to 3 drops of blood are applied to the cartridge until filled as indicated, and then sealed (see images below).
e. The sealed cartridge is inserted and pushed into the Handheld port until it clicks into place.
f. Results are reviewed after about 2-3 min when the test is completed.
g. Approximately 1 wk after initial blood collection, mice are fasted overnight to measure fasting glucose level.
h. Fasting glucose is measured using a handheld glucometer (see image below, right panel) according to manufacturer's instruction. Results obtained from whole blood sample is reported to be 10-15% lower than standard laboratory serum/plasma glucose results.
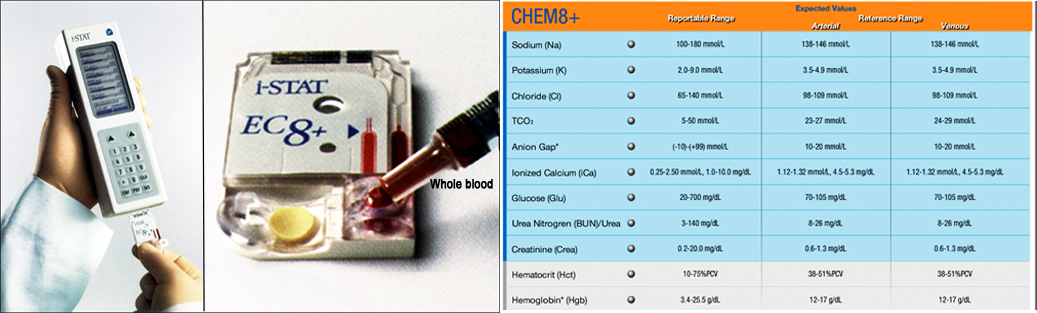
i-STAT handheld and handheld glucometerX. Procedure for necropsy and morphometry
a. One week after final blood collection, mice are fasted overnight, euthanized, and dissected for measurements of morphometric phenotypes and collection of tissue samples.
b. The presence of any obvious pathologies (e.g., spontaneous neoplasms) and all subsequent metrics are recorded in an established MouseTrack database.
c. Each mouse is placed on a scale to collect individual body weight.
d. Using a ruler tail length is obtained and recorded from the base to the tip of the tail.
e. The head is transected between C1 and C2 and the calvarium carefully dissected to expose the brain en-mass and removed for weighing.
f. The remaining carcass is dissected at midline, from the thorax to the pelvic symphysis, to expose the major organs.
g. Heart is dissected from its root in the thoracic cavity and weighed.
h. Spleen and both pair of kidneys are isolated from the abdominal viscera and weighed. Kidney volume is assessed.
i. Remaining visceral organs are removed en mass from the root of the mesentery, and the pubic symphysis is separated to expose the reproductive organs.
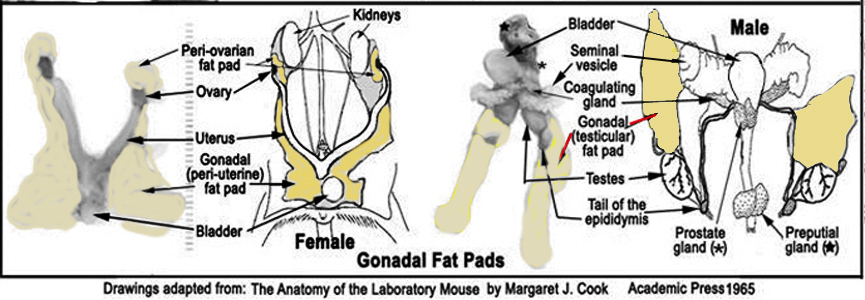
j. The perigonadal fat pads are dissected and weighed (see illustration below from Reed1). To accomplish this in females, the entire reproductive tract is dissected off the pelvic cavity.
k. In males both testes are dissected and weighed following the removal of the perigonadal fat pads for weighing.
l. The hind axial skeleton (hind limbs, pelvis, lumbar spine) and attached muscle) are placed in 95% ethanol for a minimum period of 14 days.
m. The femurs are then isolated from the musculature, and periosteal circumference is measured at the exact midshaft of the femur (Donahue2).
CC: Collaborative Cross
Founder strains: five common inbred (A/J, C57BL/6J, 129S1/SvImJ, NOD/ShiLtJ, NZO/HlLtJ) and three wild-derived inbred (CAST/EiJ, PWK/PhJ, WSB/EiJ) mice
Collaborative Cross: is an emerging mouse recombinant inbred (RI) panel that was designed to extend the available mouse genetic reference populations (Philip et al. 2012) by combining the genomes of eight genetically and phenotypically diverse strains through randomized breeding design that systematically outcrosses eight founder strains, followed by inbreeding to obtain new recombinant inbred strains.
Data collected by investigator
blood chemistry
- sodium (blood Na)
- potassium (blood K)
- chloride (blood Cl)
- calcium (blood ionized Ca)
- anion gap concentration
- carbon dioxide (blood CO2)
- glucose (blood GLU)
- glucose (blood GLU, overnight fast)
- blood urea nitrogen (BUN)
- creatinine (blood)
blood cell counts
- white blood cell count (WBC; units per volume x 103)
- lymphocyte count (LYM; units per volume x 103)
- lymphocyte differential (LYM; percent of viable WBC)
- monocyte count (MONO; units per volume x 103)
- monocyte differential (MONO; percent of viable WBC)
- granulocyte count (GRA; units per volume x 103)
- granulocyte differential (GRA; percent of total viable WBC)
- eosinophil differential (EOS; percent of viable WBC)
- red blood cell count (RBC; units per volume x 106)
blood erythrocyte and platelet parameters
- mean RBC corpuscular volume (MCV)
- RBC corpuscular distribution width (RDW)
- mean RBC corpuscular hemoglobin (MCH)
- mean RBC corpuscular hemoglobin concentration (MCHC)
- hemoglobin (HGB)
- hematocrit (HCT)
- platelet count (PLT; units per volume x 103)
- mean platelet volume (MPV)
reproduction
- number of mice born in first litter
- parental age at first litter
- time from mate to first litter
wildness rating
- capture
- hold
- total
open field behavior
- locomotor activity
- first 3 min
- total
- transitions
- center occupancy
- first min
- total
- distance
- percent
- thigmotaxis
- latency
- duration
- anxiety-related behaviors
- immobility
- defecation
- urination
light-dark arena behavior
- light zone occupancy
- duration
- distance
- percent
- anxiety-related behaviors
- latency
- transitions
- defecation
- urination
sleep patterns
- baseline
- total
- dark period
- after sleep deprivation
- total
- dark period
- peak activity
nociception
- latency of nociceptive response to hot plate (54°C), 30 s test
- latency of nociceptive response to pressure on the tail, 60 s test
morphometrics
- body weight
- body length
- tail length
- brain weight
- heart weight
- kidney length
- kidney volume
- kidney weight
- spleen weight
- testicular weight
- gonadal fat pad weight
- periosteal (outer) circumference, femoral mid-diaphysis