Peters4 project protocol
Aging study: Blood hematology in 30 inbred strains of mice (2007)
Peters LLWith: Schultz D, Godfrey D
Project protocol — Contents
Workflow and sampling
Equipment
Reagents, supplies, and solutions
Procedure for CBC with differentials using ADVIA system
Data
References
Workflow
Body weight measurement Blood collection; samples are kept at 24°C (for <3 h) until testing Hematology samples are run on the ADVIA 120 Hematology Analyzer Bayer ADVIA 120 Hematology Analyzer (Bayer Diagnostic Division (Siemens), Tarrytown, NY, USA.
More equipment information
ADVIA® 120 Hematology System Product Features
Panel A shows the Bayer ADVIA 120 hematology analyzer system. Panel B shows the internal layout of the ADVIA, and the various reagents used in the system. Panel C shows the hematology setup, equipped with a computer and a printer. Panel D shows a typical computer screen-output.
- Microhematocrit tubes: heparinized, 75 mm, 75 µL volume size (Fisherbrand®)
- Blood collection tubes: Microtainer® tube containing lyophilized 2EDTA (BD (Becton-Dickinson) Diagnostics, Franklin Lakes, NJ) or (Fisher Catalogue # 36-5974)
- Toothpicks
- Acclimation to test conditions
- In general all mice are brought into the procedure room and are tested within 1h.
Procedure: CBC with differential and reticulocyte count using Bayer ADVIA 120 Hematology Analyzer
I. Standard blood collection
a. Mice at 6-7.5, 12-13.5, and 18-19.5 months of age are fasted for 4 hrs (7:30 to 11:30 am) before they are bled.
b. Body weight measurements are recorded just before blood samples are drawn.
c. 270 µL of blood sample (about 11 drops plus one capillary tube) is collected via a retro-orbital bleed using a heparin-coated capillary tube directly into BD Microtainer tube coated with lyophilized K2EDTA.
d. The freshly drawn blood is quickly and gently mixed with the anticoagulant using a toothpick. Vortexing or inverting the sample is not done in order to preserve the proper ratio of anticoagulant to blood and also to preserve the integrity of the Red Blood Cells.
e. Whole blood specimens are kept at room temperature until analysis, however, anticipated delay of greater than 3 hrs required sample refrigeration, and subsequent return to room temperature before testingII. Blood cell count with differential using Bayer ADVIA 120 system
a. Approximately 210 µL of un-clotted whole blood is transferred to a 2nd microtainer and is analyzed using the ADVIA 120 hematology system according to manufacturer's protocol.
b. For additional information see The Aging Center at The Jackson Laboratory.Data collected by investigator
Submitted measurements: White blood cell count (WBC), red blood cell count (RBC), neutrophil count (NEUT), lymphocyte count (LYMPH), monocyte count (MONO), eosinophil count (EOS), basophil count (BASO), large unstained cell (LUC), and reticulocytes (Retic) count, % neutrophil, % lymphocytes, % monocytes, % eosinophils, % LUC, % basophils, mean platelet volume (MPV), and platelet (PLT) count; red cell parameters including hematocrit (HCT), hemoglobin (HGB), mean RBC corpuscular hemoglobin content (MCH) and concentration (MCHC), mean RBC corpuscular volume (MCV), % reticulocytes (Retic), and reticulocyte corpuscular hemoglobin content (CHr) for 6, 12, 18, and 24 months old mice.
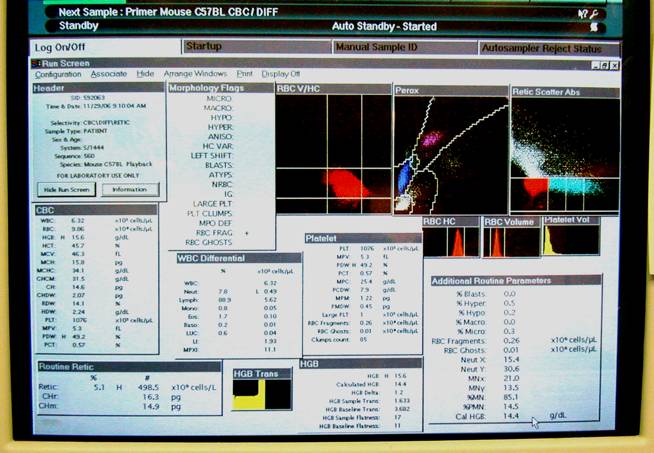
This figure illustrates a close-up display of the computer screen result in Panel D above.
References
Peters LL, Cheever EM, Ellis HR, Magnani PA, Svenson KL, Von Smith R, Bogue MA. Large-scale, high-throughput screening for coagulation and hematologic phenotypes in mice. Physiol Genomics. 2002 Dec 3;11(3):185-93.
PubMed 12419856 Svenson KL, Von Smith R, Magnani PA, Suetin HR, Paigen B, Naggert JK, Li R, Churchill GA, Peters LL. Multiple trait measurements in 43 inbred mouse strains capture the phenotypic diversity characteristic of human populations. J Appl Physiol. 2007 Jun;102(6):2369-78. Epub 2007 Feb 22.
PubMed 17317875
