Threadgill2 project protocol
Drug study: Acetaminophen pharmakokinetics and drug-induced liver injury in males of 5 inbred strains of mice (2005)
Threadgill DW, Harrill AWith: Ross PK, Rusyn I
Project protocol — Contents
Workflow and sampling
Equipment, supplies, software
Reagents, solutions
Procedure: Acetaminophen administration
Procedure: Blood collection and sample processing
Procedure: Blood serum chemistry
Procedure: Acetaminophen and metabolite quantification
Procedure: Liver glutathione quantification
Procedure: Histopathology to assess liver necrosis
Procedure: Nitrotyrosine immunostaining to assess hepatocyte injury
Definitions and Abbreviations
Data
ReferencesWorkflow
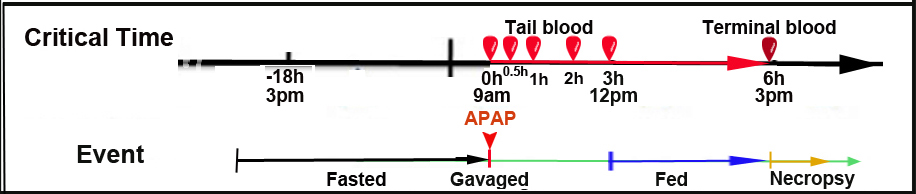
Mice fasted 18h before dosing Mice treated with APAP or vehicle via intragastric (i.g.) route Tail blood collected at designated timepoints (Fig. 1) Feed provided 3h post-treatment Mice sacrificed 6h post-treatment; terminal blood and liver samples collected Blood serum chemistry Liver glutathione quantification Liver histopathology and immunostaining See Abbreviations
Figure 1. Procedural timeline; see also study design (not drawn to scale).
- Balance scale
- Gavaging needles for mice
- Syringes (3 mL) and needles (G19)
- Dissecting tools
- Scalpel blades (#11)
- Spectrophotometer
- Centrifuge, tabletop
- Microtome for histology
- Tissue homogenizer
- High-performance liquid chromatography (HPLC) system with fluorescence detection
- pH meter
- Microscope with 40x and 100x objectives and image capture capability
- WinNonLin analysis software (Pharsight, Mountain View, CA)
- Image Pro Plus software (version 5.1; Silver Spring, MD)
- Acetaminophen (APAP; Sigma-Aldrich, St. Louis, MO)
- Methyl 2-hydroxyethyl cellulose (vehicle) (Sigma-Aldrich, St. Louis, MO)
- Nembutal (anesthesia) (Abbott Laboratories, Chicago, IL)
- 10% phosphate buffered formalin (Sigma-Aldrich, St. Louis, MO)
- Hematoxylin and Eosin stain (H & E)
- Primary antibody 1:200 nitrotyrosine (Molecular Probes, Eugene, OR)
- Dako EnVision system horseradish peroxidase (HRP) (Dako Cytomation, Carpinteria, CA)
- Alanine aminotransferase Reagent (Thermo Fisher Scientific, Waltham, MA)
- ApoGSH Glutathione Colorimetric kit (BioVision, Mountain View, CA)
- Sulfosalicylic acid
- 3-acetaminophenol (internal standard) (Sigma-Aldrich, St. Louis, MO)
- Acetonitrile
- Sodium sulfate
- Potassium phosphate
- 70% ethanol for cleaning and disinfection
Procedure: Acetaminophen administration and study design
a. Mice are singly housed throughout the duration of the experiment.
b. Mice are fasted for 18h and then weighed to determine amount of acetaminophen (APAP) needed for doses of 50 or 300 mg per kg body weight.
c. Mice are administered either vehicle (0.5% methyl 2-hydroxyethyl cellulose) or APAP intragastrically in a dosing volume of 10 mL/kg body weight (see details below for gavaging technique).
d. Blood is collected immediately and at four additional timepoints (t=0, 0.5, 1, 2 and 3h). Note: true baseline (pre-treatment) is not collected in this series (see Timeline, Fig. 1).
e. Ad libitum feeding is resumed 3h after treatment. Mice are sacrificed at 6h post-treatment; terminal blood is collected and livers are harvested (see below).Oral gavaging technique
Gavaging is used to deliver a specified volume of material directly into the stomach. Only specialized, commercially available gavage needles for mice are used.
a. The syringe is first filled with the appropriate volume of material and set aside.
b. Mice are gently restrained by the scruff, and the tip of the gavaging needle is placed in the mouth and gently pushed past the back of the tongue (the tip of the needle should slide easily down the esophagus; any resistance encountered requires the removal of the needle and reinsertion).
c. The pre-loaded syringe is attached to the needle, and material is delivered intragastrically (injury to surrounding tissues is avoided by carefully holding the syringe and preventing aspiration).Procedure: Blood collection and sample processing
a. Mice are pre-warmed under a heat source before blood collection.
b. Mice are placed on a restrainer and the tail is disinfected with 70% ethanol.
c. Using a #11 scalpel blade, the lateral tail vein is nicked (subsequent sampling is done at progressively more proximal locations along the tail). Any attempt to squeeze the tail or to "milk" blood from the tail is avoided since damage to tissues can contaminate the blood sample with tissue fluids.
d. Blood samples (5 x 50 µL) are collected from the tail vein at specified timepoints (Fig. 1) using serum separation tubes.
f. Feed is provided immediately following the last blood collection. At 6h post-treatment, mice are administered Nembutal at a dose of 100 mg/kg body weight i.p. and sacrificed.
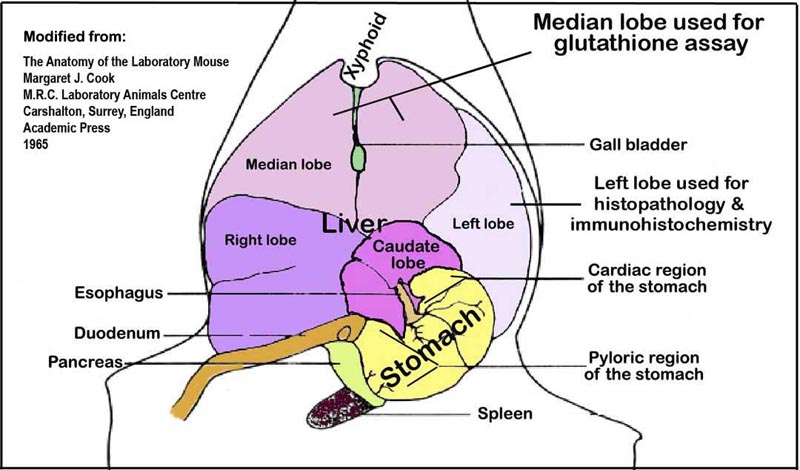
g. Terminal blood is collected from the vena cava using a 3 mL syringe and G19 needle; serum is processed (see below). Livers are quickly excised following exsanguination. Left lobes (Fig. 2) are snap-frozen in liquid nitrogen and stored at -80°C for glutathione assays (see below). Remaining liver tissue is fixed in 10% phosphate buffered formalin for histology and immunohistochemical analyses (see below).
h. Blood is transferred into a pre-labeled (no anti-coagulant) Eppendorf tube and allowed to clot at room temperature for at least 30 min.
i. Serum is separated from blood cells using a table top centrifuge at a speed of 14,000 rpm for 5 min.
j. The serum layer is pipetted carefully without disturbing the packed blood cell layer below and then transferred into a fresh tube and stored at -20°C until serum chemistry and HPLC analysis (see below).
Figure 2. An illustration of the liver lobes and their orientation within the abdominal cavity viewed ventro-dorsally.Procedure: Blood serum chemistry
a. An aliquot of frozen serum is thawed to room temperature before alanine aminotransferase (ALT) quantification.
b. ALT is quantified using a kinetic method in which the rate of oxidation of NADH by lactate dehydrogenase is measured at 340 nm wavelength using a spectrophotometer, according to manufacturer’s instructions.Procedure: Acetaminophen and metabolite quantification
a. Quantification of serum acetaminophen (APAP) and its major metabolites, glucuronide and sulfate conjugates (APAP-G and APAP-S), is accomplished using 25 µL of serum sample and a reversed-phased HPLC assay with UV detection at 254 nm wavelength.
b. The HPLC assay is run with a mobile phase consisting of 5% acetonitrile and 95% 5 mM sodium sulfate, 20 mM potassium phosphate buffer (pH=3.2) and a flow rate of 1.2 mL/min.
c. APAP, APAP-G, APAP-S, and the internal standard (3-acetaminophenol) are measured after 3.6, 5.2, 7 and 11 min retention times, respectively.
d. For each sample, the area under the curve (AUC) is calculated by using noncompartmental analysis using WinNonLin software.Procedure: Liver glutathione quantification
a. Reduced and total glutathione in liver are determined using the ApoGSH Glutathione Colorimetric kit.
b. Snap-frozen liver samples (50 mg) from the left liver lobe (Fig. 2) are homogenized in kit-supplied Glutathione Buffer™ and centrifuged for 10 min at 8000 x g following the addition of 100 µL 5% sulfosalicylic acid.
c. The supernatant is then assayed for reduced and total glutathione according to the manufacturer’s protocol.
d. Glutathione levels are quantified using the pseudo-end point method based on a generated standard curve.Procedure: Histopathology to assess liver necrosis
a. Formalin-fixed liver specimens are embedded in paraffin wax, cut to a thickness of 5 µm sections, and applied to microscope slides in duplicate.
b. One slide is stained with hematoxylin and eosin (H&E), and the other is saved for nitrotyrosine staining (see below).
c. Liver necrosis is scored by a point counting technique on the H&E slide, where a grid with 100 evenly spaced points is overlaid on a printed image of a liver section taken at 100X magnification (Fig. 3).
d. Liver necrosis is expressed as a percentage of the total area.
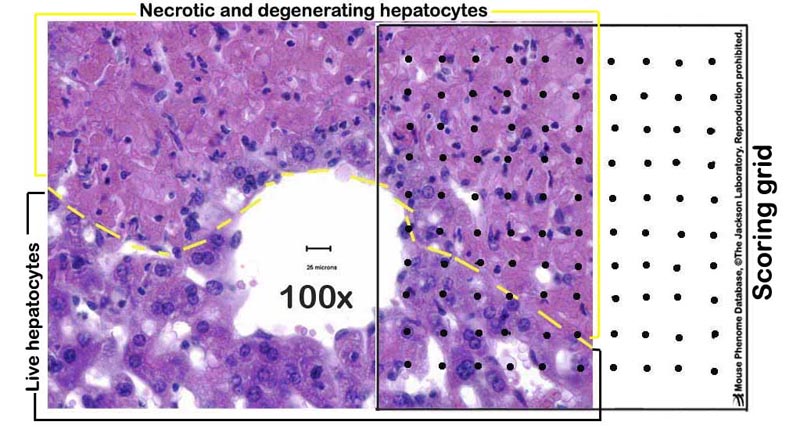
Figure 3. A photomicrograph example of a partially necrotic liver overlaid with a scoring grid.Procedure: Nitrotyrosine immunostaining to assess hepatocyte injury
a. Immunostaining is performed on the remaining slide using the Dako EnVision system HRP with primary antibody to nitrotyrosine (1:200 dilution) and counter stained with hematoxylin.
b. Slides are processed in parallel on the same day for quantitative and comparable measurements using Image Pro Plus software.
c. The area that is stained is recorded as a percentage of the total area assayed; determined from five areas per slide (at 400X magnification).APAP (acetaminophen, or paracetamol, or N-acetyl-p-aminophenol): an analgesic and antipyretic drug, commonly known by trade names Tylenol and Panadol
Percent reduced glutathione = [reduced GSH ÷ total GSH] *100
Percent liver necrosis = [number of evenly spaced overlaid points within necrotic areas of the section ÷ total number of points within the tissue section] x 100
Nitrotyrosine (marker of hepatocyte injury): an indicator of the production of nitric oxide, cell damage, and inflammation. Hepatocyte injury is expressed as a percentage of total area that stains positively with antibodies for nitrotyrosine.
alanine aminotransferase glutathione in oxidized state N-acetyl-p-aminophenol (acetaminophen, or paracetamal) hematoxylin and eosin APAP-glucuronide high performance liquid chromatography APAP-sulfate nicotinamide adenine dinucleotide area under curve drug treatment (dose) glutathione (GSH usually refers to the reduced form of glutathione) Data collected by investigator
- Serum ALT
- Serum APAP and metabolites (APAP-S and APAP-G)
- Liver total and reduced glutathione
- Percent liver necrosis
- Percent nitrotyrosine staining (marker of hepatocyte injury)