Jaxwest2:
Neoplasia induction in NOD.CB17-Prkdcscid/J female mice by passage of live tumor cells (HT29) (2007)
Jackson Laboratory
Yan Yang With: Yang Y, Rodriguez R, Braun M, Clary D
Jaxwest2 downloads
Click on "Download" link or button below, to see the available downloads for this project.
Click on "Download" link or button below, to see the available downloads for this project.
| Institutional authorship |
Jackson Laboratory
|
| Investigators |
Yan Yang The Jackson Laboratory, Bar Harbor, ME |
| Participants | Yang Y, Rodriguez R, Braun M, Clary D |
| Contact | Yan Yang yan.yang@jax.org
|
| Affiliated Center | JAX In Vivo Services |
| One-strain study | |
| MPD identifiers | Jaxwest2 MPD:257 |
| No updates/corrections. Initial release date: 06/2007. | |
|
Click above to copy-paste the entire citation for this MPD web page. |
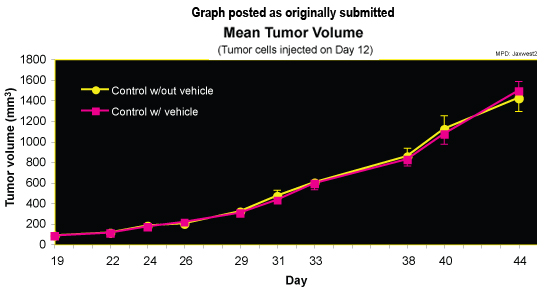
Purpose: To provide normative data for testing biologically-active compounds in immunodeficient female NOD.CB17-Prkdcscid/J strain. This strain is an excellent host for xenografts, including the human colorectal adenocarcinoma cell line, HT-29, used here for tumor induction.
By posting this data, JAX® In Vivo Services contributes to community research resources and fulfills an agreement with clients to make normative data public.
Available download: Jaxwest2.xls
X = Each of these measurements is replicated for test Day X (see protocol for more information).
The data set contains these columns: strain sex mouse_ID birth date of birth arrival date of arrival at test facility cage_code cage code at test facility HT29_inj tumor cells (HT29) injected exp_group experimental group: Control w/out vehicle; Control w/vehicle; Test Group (pre-treatment data only) bw_X body weight on Day X (after arrival) g bw_chg_X change in body weight from Day 12 to Day X % tumor_L_X tumor length on Day X mm tumor_W_X tumor width on Day X mm tumor_H_X tumor height on Day X mm tumor_vol_X tumor volume on Day X mm3 tumor_chg_X change in tumor volume from 1 wk after HT29 injection (Day 19) to Day X % tumor_wt_44 tumor weight at necropsy (Day 44) g survival_500 number of days after HT29 injection that tumor volume reached 500 mm3 n survival_1500 number of days after HT29 injection that tumor volume reached 1500 mm3 n